Products
Services
Information
About
Contact
Products
Ofni Systems provides your FDA-regulated business with software and products to assist with 21 CFR 11, Annex 11, HIPAA, and other regulatory requirements for electronic data and signatures.
Ofni Systems provides your FDA-regulated business with software and products to assist with 21 CFR 11, Annex 11, HIPAA, and other regulatory requirements for electronic data and signatures.
ExcelSafe
Part 11 Compliance For
MS Excel® Spreadsheets Simple To Use
Get spreadsheet control in a matter of hours Full Part 11 Compliance
Audit trails, electronic signatures and user security Spreadsheet Validation
We are FDA experts on spreadsheet validation Free Trial
Get a copy of Excelsafe to test today
Part 11 Compliance For
MS Excel® Spreadsheets Simple To Use
Get spreadsheet control in a matter of hours Full Part 11 Compliance
Audit trails, electronic signatures and user security Spreadsheet Validation
We are FDA experts on spreadsheet validation Free Trial
Get a copy of Excelsafe to test today
FastVal
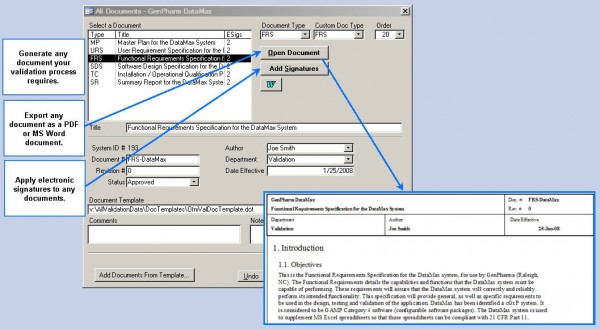
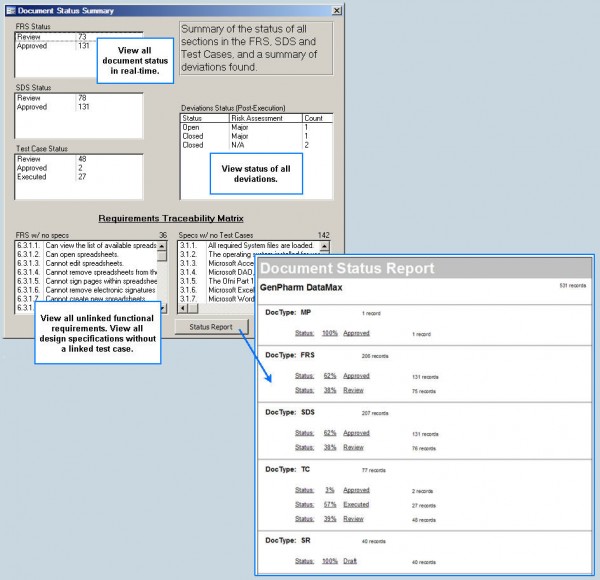
A Complete Validation Management Solution Create Documents
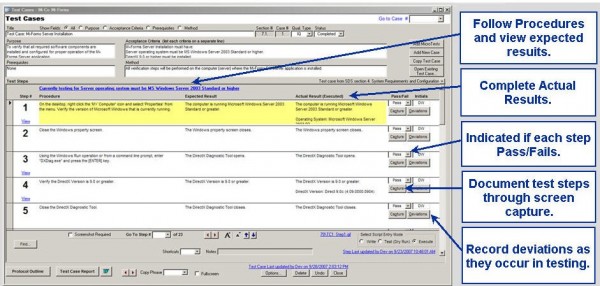
Fastval creates all your validation documents Electronic Execution
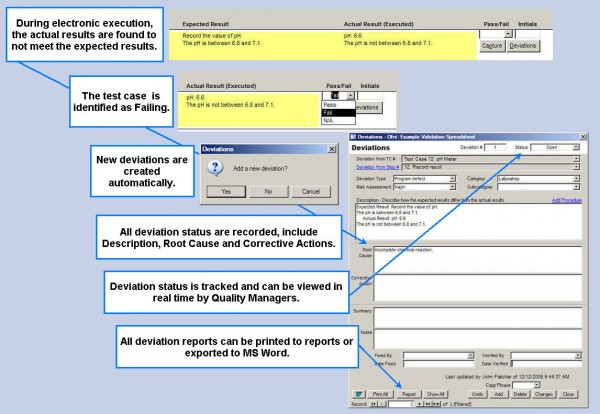
Execute validation protocols electronically Deviation Tracking
Deviation generation, tracking and management Project Management
Tools and reports to help manage people & projects
A Complete Validation Management Solution Create Documents
Fastval creates all your validation documents Electronic Execution
Execute validation protocols electronically Deviation Tracking
Deviation generation, tracking and management Project Management
Tools and reports to help manage people & projects
Ofni Clinical
eCRF Data Collection Database For Clinical Trials Case Report Forms
Converts your paper CRF's into eCRF's Edit Checks
Ensure data integrity with validation rules MedWatch Reporting
Automatically generates MedWatch FDA Form 3500A Rapid Implementation
Ready to use in days, not weeks or months
eCRF Data Collection Database For Clinical Trials Case Report Forms
Converts your paper CRF's into eCRF's Edit Checks
Ensure data integrity with validation rules MedWatch Reporting
Automatically generates MedWatch FDA Form 3500A Rapid Implementation
Ready to use in days, not weeks or months
Part 11 Toolkit
Makes Any MS Access® Database Part 11 Compliant Audit Trails
Track all changes made in a database Electronic Signatures
Secure data with electronic signatures Database Validation
Let us validate your database for you Free Trial
Try the Part 11 Toolkit in your own database
Makes Any MS Access® Database Part 11 Compliant Audit Trails
Track all changes made in a database Electronic Signatures
Secure data with electronic signatures Database Validation
Let us validate your database for you Free Trial
Try the Part 11 Toolkit in your own database
Part 11 Advisor
Part 11 Auditing, Remediation and Training Tool Starting Gap Analysis
Facilitates the process of starting gap analysis Assessments
Collect and review data to identify Part 11 gaps Long-Term Tracking
Track the completion of your corrective action plan On-site Implementation
Hands on training on 21 CFR Part 11
Part 11 Auditing, Remediation and Training Tool Starting Gap Analysis
Facilitates the process of starting gap analysis Assessments
Collect and review data to identify Part 11 gaps Long-Term Tracking
Track the completion of your corrective action plan On-site Implementation
Hands on training on 21 CFR Part 11
Information
Ofni Systems is committed to assisting organizations with electronic records compliance, such as 21 CFR Part 11 and Annex 11. Let Ofni Systems make knowledge management simple for you.
Ofni Systems is committed to assisting organizations with electronic records compliance, such as 21 CFR Part 11 and Annex 11. Let Ofni Systems make knowledge management simple for you.
21 CFR Part 11
A complete set of resources for 21 CFR Part 11 21 CFR 11.10(a)
Validation of computer systems 21 CFR 11.10(b)
Accurate Generation of Records 21 CFR 11.10(c)
Protection of Records 21 CFR 11.10(d)
Limited System Access 21 CFR 11.10(e)
Audit Trails 21 CFR 11.10(f)
Operational System Checks 21 CFR 11.10(g)
Authority Checks 21 CFR 11.10(h)
Input Checks 21 CFR 11.10(i)
Education, Training and Experience 21 CFR 11.10(j)
Policies for Electronic Signatures 21 CFR 11.10(k)
Document Control Part 11 FAQ
Validation of computer systems
A complete set of resources for 21 CFR Part 11 21 CFR 11.10(a)
Validation of computer systems 21 CFR 11.10(b)
Accurate Generation of Records 21 CFR 11.10(c)
Protection of Records 21 CFR 11.10(d)
Limited System Access 21 CFR 11.10(e)
Audit Trails 21 CFR 11.10(f)
Operational System Checks 21 CFR 11.10(g)
Authority Checks 21 CFR 11.10(h)
Input Checks 21 CFR 11.10(i)
Education, Training and Experience 21 CFR 11.10(j)
Policies for Electronic Signatures 21 CFR 11.10(k)
Document Control Part 11 FAQ
Validation of computer systems
Validation Resources
On selected validation documents Validation Planning
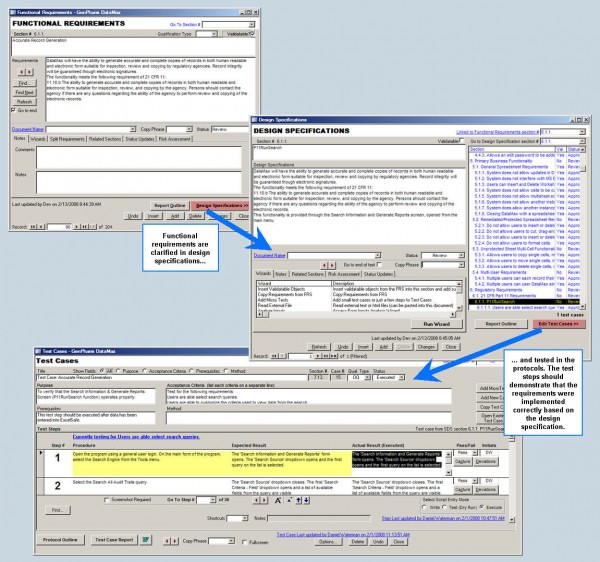
Define the scope and goals of a validation project. Requirement Gathering
Operations and activities that a system must be able to perform. Design Specification
Design Specifications describe how a system performs the requirements Installation Qualification
Verifies the proper installation and configuration of a System. Summary Report
Provides an overview of the entire validation project.
On selected validation documents Validation Planning
Define the scope and goals of a validation project. Requirement Gathering
Operations and activities that a system must be able to perform. Design Specification
Design Specifications describe how a system performs the requirements Installation Qualification
Verifies the proper installation and configuration of a System. Summary Report
Provides an overview of the entire validation project.
Other Resources
More information on FDA compliance FDA Warning Letters
Sample FDA 483 and Warning Letters Part 11 Publications
Useful 21 CFR 11 resources. Clinical Data Management
Best practices in handling data from clinical trials. Ofni Compliance Blog
Resources and information straight from the Ofni team.
More information on FDA compliance FDA Warning Letters
Sample FDA 483 and Warning Letters Part 11 Publications
Useful 21 CFR 11 resources. Clinical Data Management
Best practices in handling data from clinical trials. Ofni Compliance Blog
Resources and information straight from the Ofni team.